Recently the Pfizer research team published a paper on their Quick Building Blocks (QBB) program in ACS Medicinal Chemistry Letters. PharmaBlock is proud to be selected as one of the key vendors of building blocks (BB) working with AMS team to support this program.
In this article, the researchers disclosed a novel business partnership that achieves the goals of just-in-time, economical access to commercial BB that increases chemical space coverage and accelerates the synthesis of new drug candidates. They said they have observed multiple advantages from the utilization of the QBB process at Pfizer and believe that this model can be of benefit to companies of all sizes that are engaged in drug discovery by reducing cost, increasing diversity of analog molecules in a time-conscious manner, and reducing BB inventory.

Researchers mentioned the key criteria to select vendors for external Just-in-Time building blocks inventory model:
1. vendors that have large numbers of desired BB in stock, with inventory in locations that could enable fast delivery to Pfizer's synthesis sites;
2. vendors that provide BB quantities needed for the current synthetic scheme;
3. vendors that be able to establish a simple set price/unit measure to shorten quoting processes and provide cost transparency for each BB;
4. vendors that be able to manage the reliability of their BB inventory to support >80% on time order fulfillment;
5. compound aggregators support logistics;
The researchers identified the vendors with significant supplies of BB who meet the criteria (Table 1, Points 1−4), with Aldrich Market Select (AMS) agreeing to play the key role of BB aggregator (Table 1, Pt. 5). PharmaBlock is honored to be involved as a key vendor of BB and work with AMS to participate in this program, and it is aligned with our commitment to supporting acceleration of drug R&D.
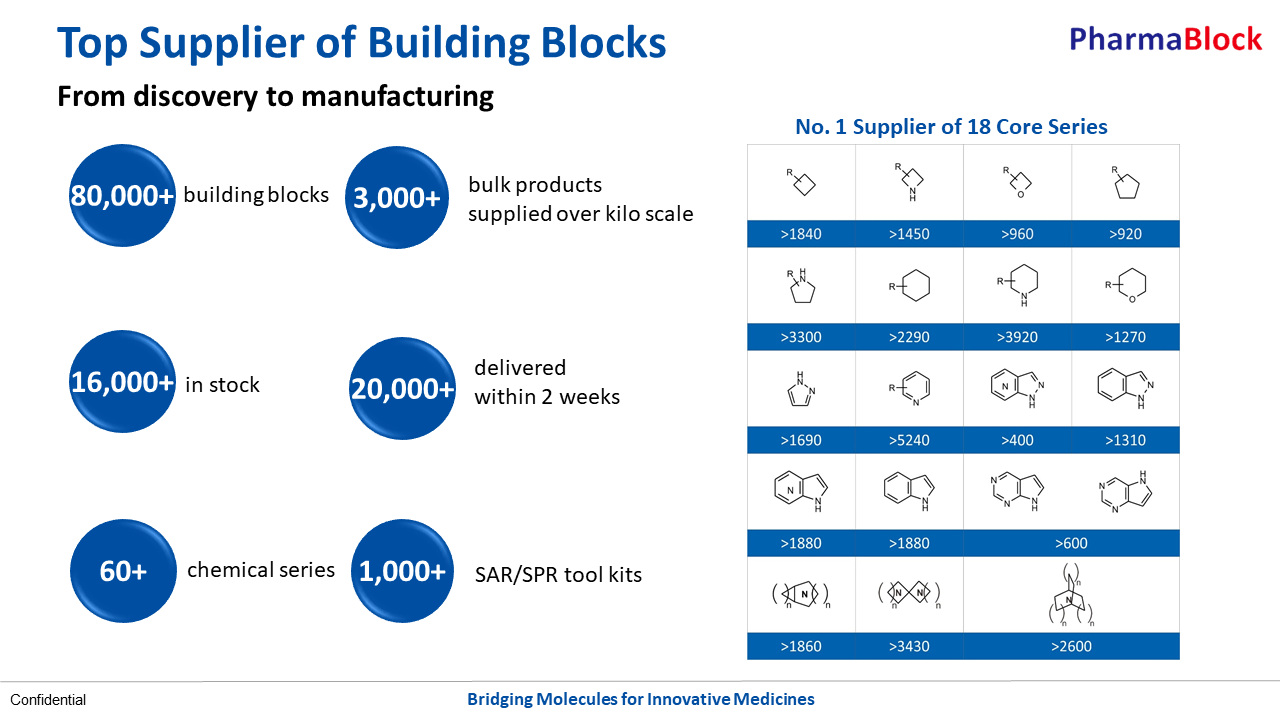
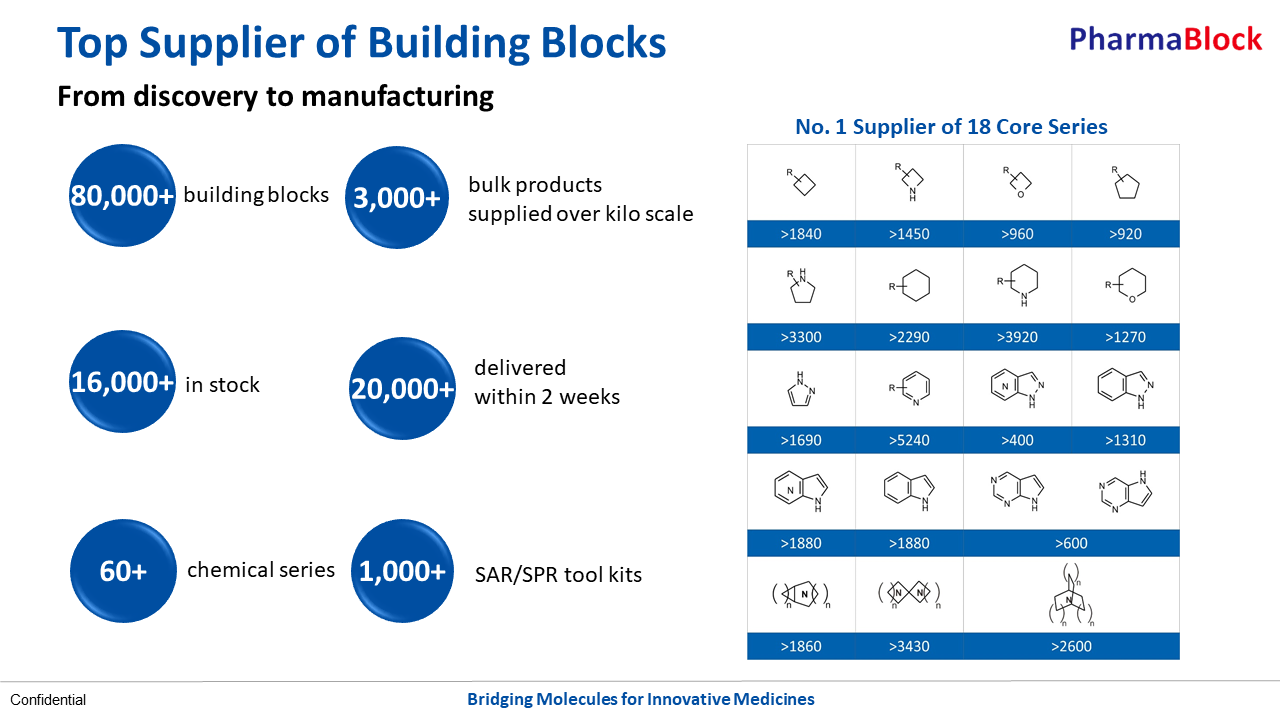
As the top supplier of building blocks around the world, PharmaBlock is recognized for our outstanding capability in the design, synthesis, and supply of novel building blocks. By monitoring the latest research in medicinal chemistry and studying marketed and clinical-stage drugs, our chemists keep expanding the product portfolio. These building blocks stocked both in China and US have been increasingly adopted in new molecules for drug discovery.
With more drug R&D projects applying the company’s products advance from discovery to preclinical and clinical research, even into commercialization, PharmaBlock moved on to build up top notch process R&D capabilities, and is providing scale-up synthesis and manufacturing of building blocks and GMP/non-GMP intermediates.The company has two manufacturing sites at Shangyu, Zhejiang Province and Dezhou, Shandong Province, with around 250 cu.meter combined reactor volume, ranging from 200L to 6,300L of reactor size. The company is now committed to a more comprehensive and long-term service to valued customers.

